 
He expected that the seventh of this series would end a 32-element period which contained elements like thorium and uranium and have an atomic weight of 292, close to the 294 now known for the first and only confirmed isotope of oganesson. Danish chemist Hans Peter Jørgen Julius Thomsen predicted in April 1895, the year after the discovery of argon, that there was a whole group of chemically unreactive gases similar to argon that would link the halogen and alkali metal groups. The possibility of a seventh noble gas, after helium, neon, argon, krypton, xenon, and radon, was considered almost as soon as the noble gas group was discovered. This was a placeholder name until the element was discovered and a name was given. It had a previous name given by the IUPAC called "Ununoctium" meaning "one-one-eight" in Latin. It was formally named on 28 November 2016. In December 2015, it was recognized as one of four new elements by the Joint Working Party of the international scientific people at IUPAC and IUPAP. It was first created in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia by a joint team of Russian and American scientists. The element is named in honor of Yuri Oganessian. The radioactive oganesson atom is very unstable, and since 2005, only five (possibly six) atoms of the isotope Oganesson-294 have been created. Oganesson has the highest atomic number and highest atomic mass of all known elements. Xenon hexafluoride on partial hydrolysis produces compounds X & Y.Oganesson is a synthetic chemical element with symbol Og and atomic number 118.Which has trigonal bipyramidal shape ?.Lithium aluminium hydride reacts with silicon tetrachloride to form.Iodine reacts with concentrated HNO3 to yield Y along with….Electron gain enthalpy with negative sign of fluorine is.Diborane B2H6 reacts independently with O2.Chlorine water on standing loses its colour and forms.Chlorine on reaction with hot and concentrated sodium hydroxide gives. 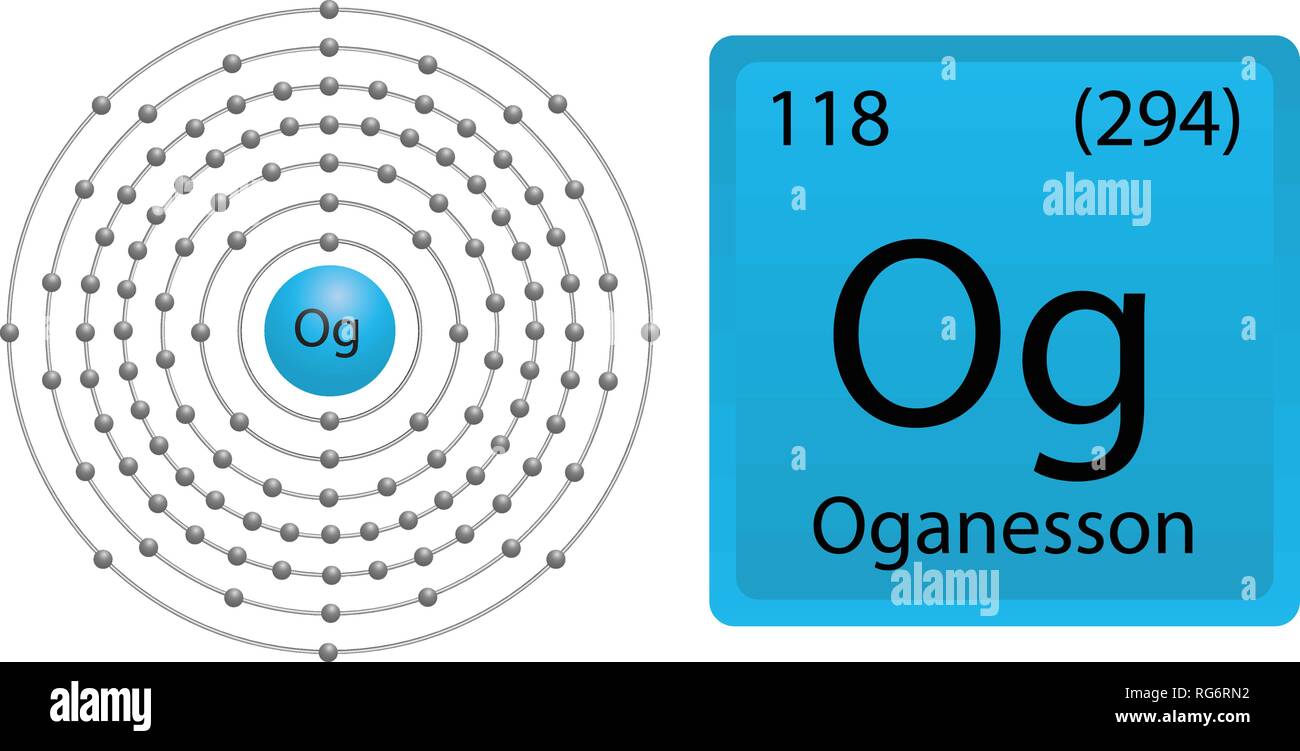
Nitrogen and Oxygen are the main components in the atmosphere but these.Among the following oxoacids, the correct decreasing order of acidic strength is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
